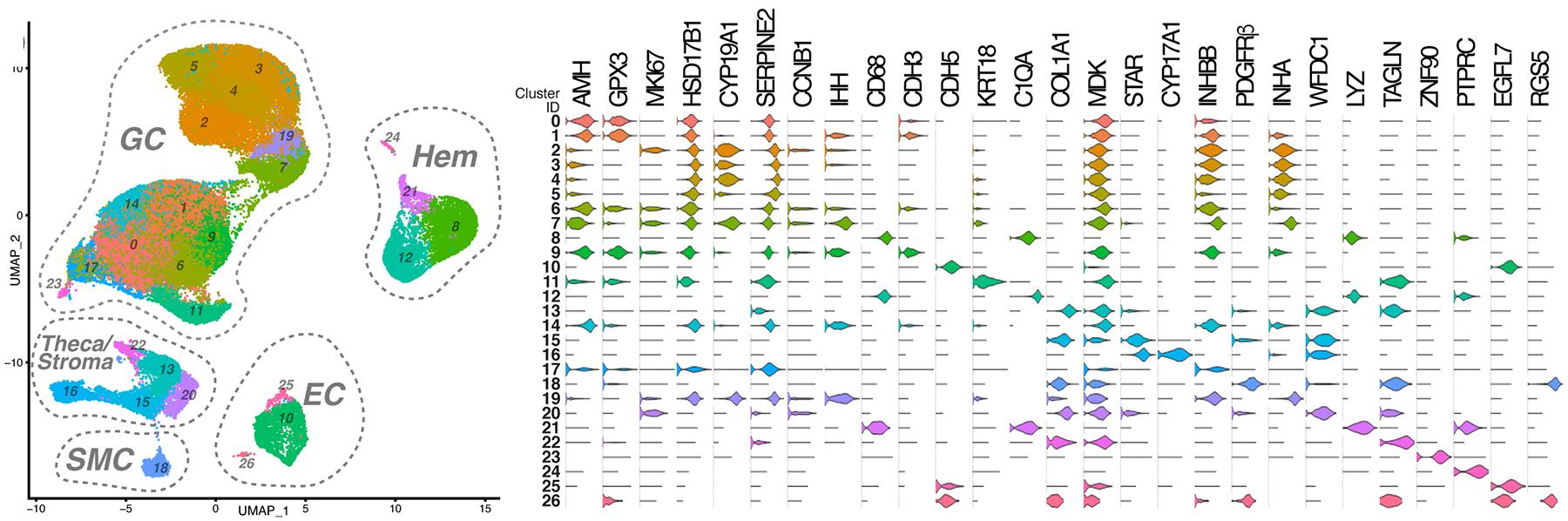
The advent of single-cell “omics” technologies has provided unprecedented insight into the heterogeneity present in cellular populations. Ovarian follicles grow from ~30 microns to ~20 millimeters over months as they mature to an ovulatory phase. This process involves the specification and expansion of multiple specialized cell types that govern metabolism, steroidogenesis, structural support, and other critical functions. In some respects, the activation, growth and maturation of an ovulatory follicle is a process more akin to embryonic organogenesis than it is to organ homeostasis. We have utilized single-cell multi-omics approaches to define the diversity of specialized cell types, as well as their origins, that are present in developing follicles. The studies have provided a transcriptional signature of follicle-resident cells that may serve as a referent in identifying the etiology of ovarian dysfunction and/or infertility, and we have used these datasets to elucidate a pathological effect of superphysiological AMH. These data also establish a phenotypic repertoire of follicle-resident cells and their molecular function that can be applied toward efforts to foster in vitro follicle growth and/or oocyte maturation.