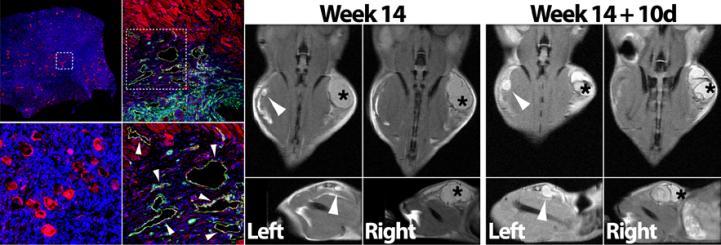
Cancer therapy has made tremendous gains in recent decades, leading to astounding increases in long-term survival, however, treatments often have a toxic effect on the gonad and can lead to infertility. Women of reproductive age routinely undergo ovarian hyperstimulation and oocyte cryopreservation to ensure that they are able to pursue family building goals once they are in remission. In girls diagnosed with cancer before puberty or for women that cannot undergo ovarian hyperstimulation for other reasons, cryopreservation, and later, auto-transplantation of ovarian tissue is the only guaranteed means of protecting their reproductive reserve. More than 200 babies have been born following ovarian tissue auto-transplantation (OTAT), but reintegration of the tissue into the body results in ischemic loss of most of its function, undermining the success of OTAT in most patients. Our lab has developed a xenotransplantation model that uses co-transplanted endothelial cells (ECs) to promote improved recovery of transplanted human ovarian tissue. This system provides a cell-based approach to improving OTAT in patients, and can also be applied to model human ovarian biology in an experimentally accessible platform. Indeed, the incorporation of ECs into the periphery of xenografted tissue provides a means of delivering paracrine factors over long-term intervals. We have used this approach to model the influence of multiple ovarian signaling effectors on human folliculogenesis.